- Atomic weight, also called relative atomic mass, ratio of the average mass of a chemical element ’s atoms to some standard. Since 1961 the standard unit of atomic mass has been one-twelfth the mass of an atom of the isotope carbon-12.
- This is a list of chemical elements, sorted by atomic mass (or most stable isotope) and color coded according to type of element.Each element's atomic number, name, element symbol, and group and period numbers on the periodic table are given. The number in parenthesis gives the uncertainty in the 'concise notation' dis given in parenthesis next to the least significant digits to which it.
- Atomic Mass of First 30 Elements - Atomic Mass of Elements is Measured with the Help of its Unit - Atomic Mass Unit. One such Unit is Equal to One-Twelfth of the Mass of a Carbon-12 Atom Which is at Rest. Learn about Atomic Mass of First 30 Elements at Byjus.
- Examples of molecular weight computations: C14O162, S34O162. Definitions of molecular mass, molecular weight, molar mass and molar weight. Molecular mass (molecular weight) is the mass of one molecule of a substance and is expressed in the unified atomic mass units (u). (1 u is equal to 1/12 the mass of one atom of carbon-12).
To find the average atomic mass, you take a certain number of atoms, find the total mass of each isotope, and then divide the total mass of all the atoms by the total number of atoms. Assume that you have, say, 10 000 atoms of carbon. Then you have 9893 atoms of 12C and 107 atoms of 13C. Mass ofl12C = 9893atoms × 12 u 1atom = 118 716 u.
| Isotope | Atomic mass (Da) | Isotopic abundance (amount fraction) |
|---|---|---|
| 12C | 12(exact) | [0.9884, 0.9904] |
| 13C | 13.003 354 835(2) | [0.0096, 0.0116] |
The 12C isotope has served since 1960 as the scale-determining reference for the definition of theunified atomic mass unit and is the basis of all atomic weights. The zero value for the delta scale usedin relative isotope-ratio measurements of carbon since the 1950s was based on a sample of fossil marine carbonate(Belemnitella Americana, Peedee Formation, Cretaceous Period, South Carolina, also known as PDB).
Atomic Mass Of C02
In 1961, the Commission recommended Ar(C) = 12.011 15(5) and in 1969 it recommended Ar(C) = 12.011(1).The larger uncertainty was assigned to include all terrestrial sources of carbon whose isotopic compositions had been measured tothat time. After the supply of PDB was exhausted, a modified delta scale was recommended for relativecarbon isotope-ratio measurements (referred to as the Vienna PDB, or VPDB scale) that yields the samezero value as the PDB scale when based on measurements of a new carbonate reference material knownas NBS 19. In 1995, the Commission recommended Ar(C) = 12.0107(8) as a result of a re-evaluationof variations in normal terrestrial materials.
Carbon Atomic Weight
Variations in the n(13C)/n(12C) ratio of terrestrial sources of carbon are caused largely by biogeochemicalreactions and physical processes. Some of the largest effects are associated with oxidation-reductionreactions including photosynthesis, such that organic substances and reduced natural gases typicallyare depleted in 13C relative to carbonate materials and the atmosphere. Differences in the degreeof 13C depletion during photosynthesis are characteristic of some groups of plants and may be passedalong to plant consumers, such that carbon isotope studies can be used to identify features of animal dietsand paleoclimates. Variations in the relative rates of organic carbon production, burial, and oxidation throughgeologic time are recorded in the isotopic compositions of sedimentary rocks. The highest reported 13C abundance is from dissolved carbonate in reduced marine sediment pore water with x(13C) = 0.011 466 andAr(C) = 12.011 50. The lowest reported 13C abundance is from crocetane recovered from the ocean bottom at cold seeps in the northern Pacific Ocean with x(13C) = 0.009 629 and Ar(C) = 12.009 66.
The radioactive 14C isotope has a half-life of 5730 a. It is introduced continuously to the near-surfaceenvironment of the earth by cosmic-ray reactions, from cosmic dust, and by nuclear technology. Itis of great interest for prehistoric dating as well as archaeological, anthropological, paleotemperature,and zoological studies. Yet, this isotope never occurs in normal carbon sources in concentrations high enoughto affect significantly the Ar(C) value. Before nuclear weapons tests, the abundance of 14C in the atmospherehad an average value of only about 10−16. It should be noted that a half-life of 5568 a (theso-called 'Libby half-life'), has been adopted by convention for calculations in geochronology.
Atomic weights of the elements 2009 by M.E. Wieser and T.B. Coplen. Pure Appl. Chem. 2011 (83) 359-396
CIAAW
Atomic Mass Of C13
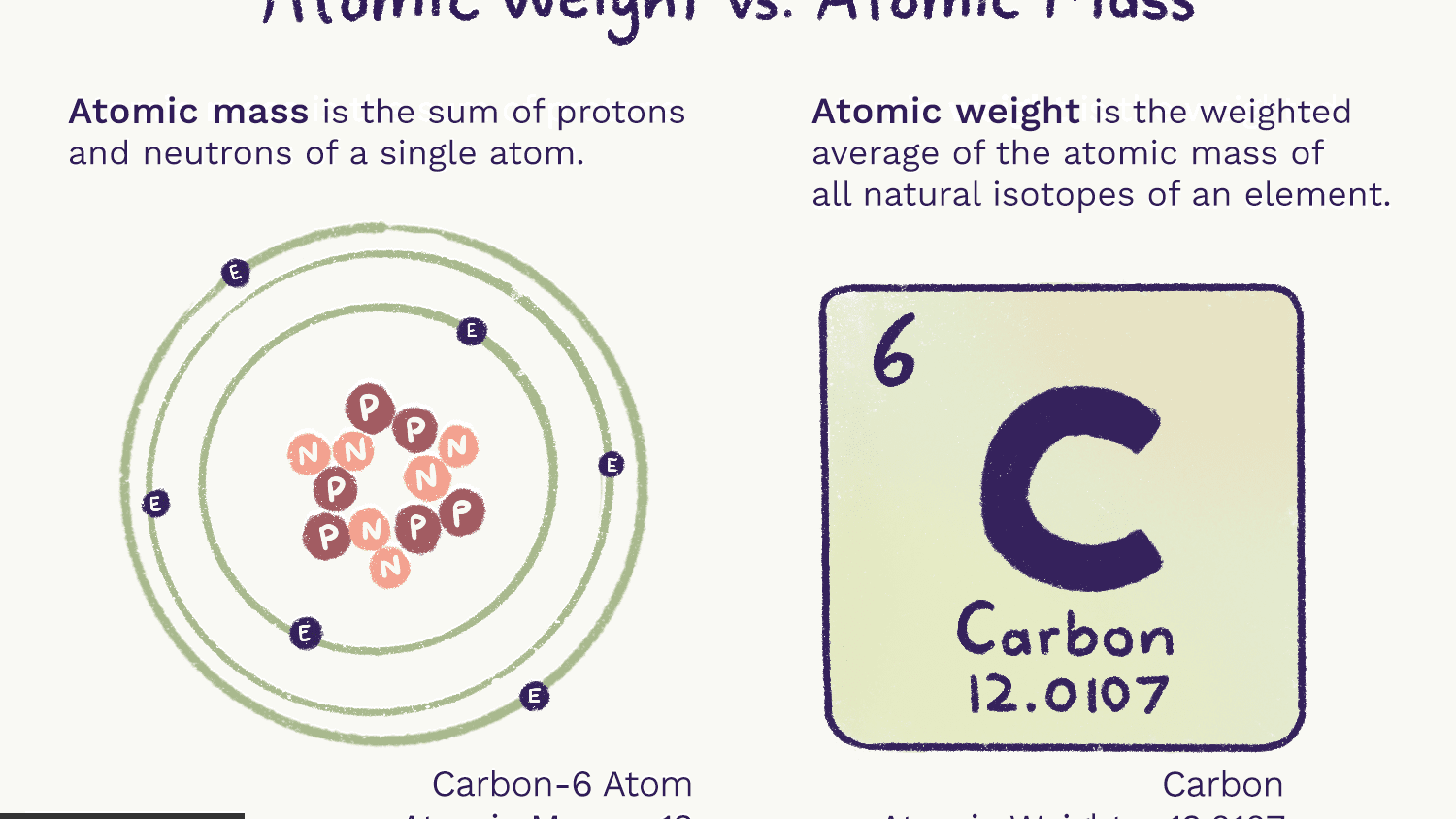
Carbon
Ar(C) = [12.0096, 12.0116] since 2009
The name derives from the Latin carbo for 'charcoal'. It was known in prehistoric times in the form ofcharcoal and soot. In 1797, the English chemist Smithson Tennant proved that diamond is pure carbon.
Ar(C) = [12.0096, 12.0116] since 2009
The name derives from the Latin carbo for 'charcoal'. It was known in prehistoric times in the form ofcharcoal and soot. In 1797, the English chemist Smithson Tennant proved that diamond is pure carbon.
Natural variations of carbon isotopic composition
Atomic Mass Of Carbon Dioxide
Isotopic reference materials of carbon.
